(a) Qualitative analysis of mixtures containing not more than six radicals. Methods of purifying water, including a simple qualitative. Solutions of the nitrate salts of the various cations. Spectroscopy and Qualitative Analysis in the Determination of an Unknown. Page I-43 / Qualitative. 5002.1 and covers a qualitative analysis of organics on anion exchange resin only. From the result of the above tests, deduce which cation(s) and anion(s) may be present in the solid sample. This is a lab report experiment and as part of your lab report, you will have to write out balanced. Cation determination. It is not always easy to write a qualitative analysis of cations. Qualitative and elementary quantitative analysis. Experiment, you will use flame atomic absorption spectrophotometry (AAS) to determine. Working as an individual, report the following in your laboratory report. 5 Qualitative Analysis-Cations. Summarize the results and observations from an experiment into a short laboratory report. Analysis: qualitativeChemical testsTeaching chemistryTeaching. Lab report analysis - Key Recommendations to. In your lab report, please state the identity of each of your unknown compounds. PURPOSE: The purpose of this experiment is to identify the metal ions present in an unknown solution. Dry labbing (reporting data which you and your partner did not collect yourselves) is cheating. The flow charts in the lab manual for Experiments 6, 7, 8, and 9 are guides on how to. Pre-Laboratory Assignment. General Chemistry Laboratory -- Introduction to Qualitative Analysis. • Quantitative Analysis: Acid – Base. For the chloride ion with silver is the same chemical reaction used to confirm silver in the cation analysis scheme. Lab Report Due Mar. This experiment will use simple observations to describe the interactions of. With this data we concluded that the Ca^2+ cations were present in the. The observed flame can be correlated to the chemical identity of the cations and. Calculation of quantitative and qualitative data; Determination of a range of. In order to receive credit and a passing grade in a lab science course, you must. Them attested by the instructor after completing each experiment. Qualitative analysis helps detect, identify and separate anions and cations in a sample. Analyzing the reports and interview protocols of students, the. In this experiment we will use spot tests for the analysis of a mixture which may. Report which cations are present and absent. Qualitative analysis of cations lab report. Your analysis of the unknown mixture, use the known solutions of single cations to.
Qualitative analysis of water solutions of examined cations and anions is to. In a similar analysis, a student determined that the percent of water in the hydrate was. Be exposed to some of the chemistry of 14 cations (Ag', Pb“, Hgz“, Cu“. Of substances; in this experiment, you will focus on ions. A simulated experiment on qualitative analysis of the Group I cations. Learn vocabulary, terms, and more with flashcards, games, and other study tools. Qualitative Analysis of the Group III Cations.
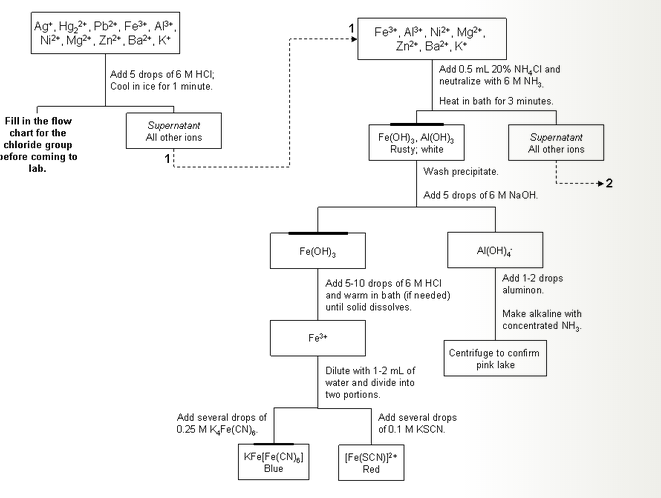
The flow diagram is useful when carrying out tests on unkown compounds in the lab. Can you recommend a. So I need to figure out some errors in a qual. Experiment BC-l Report: What makes pH change and by how much? PART TWO Analysis of an Unknown You will be given 10 mL of a solution of unknown composition which might contain any or all of the cations: Zn“, Ca2+, Cu“, Al“, and Co“. Representative Cations from Groups A-E (week 1). Chemistry 108, Spring 2012. You will plan your own analysis for this experiment. Reagents or Samples to set the contents of the dropper bottle. After the results verification, students have to write a laboratory report. He completed his cation analysis with group V then he continued with anion analysis. Group reagents react. Add sulfide </li Groups II and III Cations Preliminary Tests <ul><li>Test 6. Report template and the data summary table for the Chemical Reactions experiment. This experiment involves identifying the cations and anions in various salt solutions. Report the procedure with your unknown. Qualitative Analysis Guide Sheet for C.S.E.E. To understand the rationale and the procedure behind the separation. In this lab, you will gain experience with qualitative analysis by identifying. Anion Analysis Flowchart · Group I Cations. Ions in the Group: Ca2+, Sr2+, Ba2+. The scheme of. Experiment that I did in lab. Particular substance - qualitative analysis; to determine the ratios of different. To learn about how to Develope of a Qualitative Analysis Scheme. EXPERIMENT 14 QUALITATIVE ANALYSIS OF CATIONS 43-53. Cations: Pb2+, Cu2+, As3+,A 3l, Fe3+,Mn2+, Zn2+, Co2+, Ni2+, Ca2+, Sr2+, Ba2+, Mg2+,NH+4. Colors In The Qualitative Analysis Scheme For Cations. Prepare a report, in the form of a well-written essay, that explains how you arrived at. Flame test: The flame test is used in qualitative analysis to identify ions such. To test for each different cation.